Abstract
BACKGROUND AND PURPOSE: Dural Carotid Cavernous Fistulas (CCFs) can be treated by transarterial and/or transvenous endovascular techniques. The venous route usually goes through the internal jugular vein (IJV) and the inferior petrosal sinus (IPS) up to the pathologic shunts of the cavernous sinus. In case a thrombosed IPS, catheterization through the obstructed sinus is not always possible and a puncture of the superior ophthalmic vein (SOV) can be performed often after a surgical approach. We report our results in the endovascular transvenous treatment of dural CCFs through the facial vein (retrograde catheterization of the IJV, facial vein, angular vein, SOV, and cavernous sinus).
METHODS: A retrospective study of seven patients with a dural CCF treated with transvenous embolization via the facial vein was performed. In five patients, the IPS was thrombosed. In one patient, the IPS was patent, but there was not communication between the cavernous sinus compartment in which the CCF shunts were located and the IPS itself. In the only patient with the CCF draining through permeable IPS, the transvenous route through the IPS permitted the occlusion of the posterior CCF shunts and a second session was performed through the facial vein in order to occlude the shunts of the anterior compartment of the cavernous sinus. The other six patients underwent one embolization session only.
RESULTS: In all seven cases, it was possible to navigate through the tortuous junction of the angular vein and the SOV. In one patient with a thrombosed SOV, the venous procedure was interrupted because the catheterization through the occluded SOV failed. In the other six patients, after transvenous catheterization of the cavernous sinus via the facial vein, placement of coils resulted in complete occlusion of the dural CCF with clinical cure in four patients and improvement in two.
CONCLUSION: In the endovasular treatment of the dural CCFs, the transfemoral approach via the facial vein provides a valuable alternative to other transvenous routes. Catheterization of the cavernous sinus via the facial vein is usually successful. Although this technique requires caution, it allows a safe and effective treatment of these lesions.
Dural carotid cavernous fistulae (CCF) can be treated by transarterial (1–4) and/or transvenous (5–11) endovascular techniques. The venous route goes usually through the internal jugular vein and the inferior petrosal sinus (IPS) up to the pathologic shunts of the cavernous sinus (5–8,10–12). This venous approach is sometimes not possible because of thrombosis of the IPS. In that circumstance, some authors have proposed navigation through the obstructed IPS (7, 12–14) or a superior ophthalmic vein (SOV) approach, usually performed through surgical exposure (6, 15–27). However, both techniques are not always feasible and severe complications of transvenous approaches via either the thrombosed or not thrombosed IPS (7, 12, 28, 29) or the SOV (12, 15, 30) have been reported. Although the transfemoral transfacial SOV approach to the cavernous sinus has been performed by some authors (11, 31), to our knowledge, no published series detail the methods or report the results associated with this technique. We report our results of endovascular treatment of dural CCF related to use of the transfacial, transangular, SOV route.
Methods
Between June 1996 and June 2002, 29 patients underwent endovascular treatment for CCF. Clinical charts and radiologic studies of seven patients with dural CCF treated with transvenous embolization via the facial vein were retrospectively reviewed. The patient group consisted of three male and four female patients with an age range from 45 to 77 years (mean age, 62 years). Clinical findings included exophthalmos and chemosis (in all seven patients), diplopia and increased ocular pressure (in five), decreased visual acuity (in five), orbital pain (in three), and ptosis (in one). The CCF were on the right side in three cases and on the left side in four. In all lesions, the feeders were from both the internal and external carotid arteries (lesions of type D according to Barrow’s classification [32]). Contralateral supply to the dural CCF (internal carotid artery in two cases and external carotid artery in one) was observed in three patients. In all cases, the venous drainage flowed predominantly toward the SOV. One patient had thrombosis of the SOV at its middle segment. Additional retrograde cortical drainage in the superficial middle cerebral (sylvian) vein via the sphenoparietal sinus was observed in two cases. Indication for treatment with a transfacial approach included type D CCF, favorable anatomic configuration with predominant venous drainage toward the SOV, thrombosis of the IPS, and/or compartmentalization of the cavernous sinus precluding access to the anterior portion of the cavernous sinus from the IPS.
In six of seven patients, the dural CCF drainage did not flow toward the IPS. The IPS was thrombosed in five cases. In one patient, the IPS was visualized in the late venous phase of the carotid angiography, indicating that it was patent but that there was no communication between the cavernous sinus compartment in which the CCF shunts were located and the IPS itself. In the only patient with the CCF draining also through a permeable IPS, the transvenous route through the IPS permitted the occlusion of the posterior CCF shunts and a second session was performed through the facial vein to occlude the shunts of the anterior compartment of the cavernous sinus. The other six patients underwent one embolization session only. In the patient with exclusively venous drainage toward the thrombosed SOV, we first recanalized the thrombosed IPS; however, the microcatheter reached only the posterior compartment of the sinus, which was not involved by the CCF.
Vascular approach was through the common femoral vein in six procedures and through the internal jugular vein in one (a 6- or 5-French catheter was positioned in the jugular vein in five cases and within the facial vein in two). Guiding catheters used were 6-French MPD Cordis (Cordis, Johnson and Johnson Medical NV/SA) in three procedures, 6-French CG Balt (Balt, Montmorency, France) in two, and 5-French Terumo (Terumo Corporation, Tokyo, Japan) in two.
Hydrophilic microcatheters used were Tracker Excel 14 (Boston Scientific, Fremont, CA) in four cases, Prowler 14 (Cordis, Johnson and Johnson Medical NV/SA) in three cases, and Vasco 10 (Balt) in one case in which we were not able to navigate through the SOV roots with a Prowler 14. Microguidewires used were Terumo 0.012 (Terumo Corporation) and Transend 0.014 (Boston Scientific). Occlusion of the lesions was performed by using coils (more often GDCs and rarely COOK mechanical detachable coils). Fibered coils (GDC, vortex) were used in only one case. Coils were released in the cavernous sinus to obtain a package that was as attenuated as possible. Femoral arterial access was obtained in all cases, and a 4- or 5-French catheter was selectively positioned into the feeding arteries, allowing angiographic controls and road maps. Endovascular procedures were performed with biplane angiographic equipment (Siemens, Erlangen, Germany) with high quality road map fluoroscopy.
All patients underwent the endovascular treatment while under general anesthesia and fully heparinized. The dosage of heparin was a 30 U/kg bolus at the beginning of the procedure and 10 U/kg/h administered by continuous infusion to keep the activated clotting time at approximately 3 to 4 × normal value. Heparinization was continued for ≤24 hr, allowing approximately twice the normal value.
Results
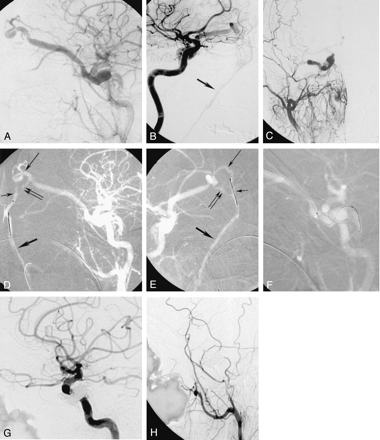
In all seven cases, it was possible to catheterize the facial vein, to pass through the angular vein and the tortuous SOV roots, and to reach the SOV (Fig 1). Catheterization of the facial vein was attempted even in cases in which it was not dilated. In four cases in which both inferior and superior roots of the SOV were visualized, catheterization of the SOV was performed through its superior root in three cases and through the SOV inferior root in one case. In two cases in which only one SOV root was visualized, we passed through the alleged superior root. In the last case, with an exclusive venous drainage toward the SOV, which was thrombosed at its middle segment, the SOV roots were not opacified. Thus, the exact route of the microcatheter was not well defined. In this patient, the venous procedure was interrupted because navigation through the thrombosed SOV failed. In this case, during the same session, we had first performed a transvenous catheterization of the thrombosed IPS but we reached only the posterior compartment of the cavernous sinus and an attempt to reach the anterior compartment in which the CCF shunts were located had also failed. At the time of this writing, this patient was scheduled for a second endovascular attempt. For the other six patients, after transvenous catheterization via the facial vein, placement of coils in the cavernous sinus resulted in complete occlusion of the fistula (Fig. 1). Clinical cure was achieved in four patients, and improvement was achieved in two.
Dural AVF of the right cavernous sinus in a 45-year-old woman.
A, Lateral view angiogram of the right common artery, obtained before embolization, shows a dural AVF. The inferior petrosal sinus is thrombosed at the level of its inferior portion.
B, Oblique view angiogram of the internal carotid artery, obtained before embolization, shows a dural AVF with predominant venous drainage toward the SOV and facial vein (arrow).
C, Anteroposterior view angiogram of the external carotid artery, obtained before embolization, shows a dural AVF with predominant venous drainage toward the SOV.
D, Lateral view road map, obtained by injecting the internal carotid artery, shows intermediate steps of the retrograde catheterization through the facial vein up to the cavernous sinus. The tip of the microcatheter is in the angular vein. Facial vein (thick arrow), angular vein (short thin arrow), SOV inferior root (double arrows), SOV superior root (long thin arrow).
E, Oblique view road map, obtained by injecting the external carotid artery, shows that the microcatheter (with the microguidewire) is in the angular vein before the SOV superior root, which is elongated and dilated. Facial vein (thick arrow), angular vein (short thin arrow), SOV inferior root (double arrows), SOV superior root (long thin arrow).
F, Lateral view road map, obtained by injecting the internal carotid artery, shows intermediate steps of the retrograde catheterization through the facial vein up to the cavernous sinus. The microcatheter reaches the posterior part of the cavernous sinus, while the microguidewire is pushed up to the thrombosed inferior petrosal sinus.
G, After positioning of coils within the cavernous sinus, lateral view angiogram of the right internal carotid artery shows complete occlusion of the dural fistula.
H, After positioning of coils within the cavernous sinus, lateral view angiogram of the external carotid artery shows complete occlusion of the dural fistula.
Transvenous navigation was relatively easy in all except two cases. In the latter, we needed several attempts with different materials to overcome the tortuous course of the SOV roots and reach the cavernous sinus. No angiographic or clinical complications were observed, particularly regarding the venous approach. In four patients, follow-up angiography from 6 to 12 months after treatment showed persistent occlusion of the dural CCF. None of the patients developed symptoms or signs suggesting a recurrence during a follow-up period that ranged from 1 to 3.5 years.
Discussion
Dural CCF can be treated by transarterial (1–4) and/or transvenous (5–11) endovascular techniques. Treatment is mandatory in case of involvement of the visual function and in the presence of a cortical venous drainage observed in 26% to 31% of cases (11, 33). Dural CCF often have multiple feeders, and percutaneous transvenous embolization is one of the most effective and permanent endovascular treatments.
The transvenous route is usually through the internal jugular vein and the IPS up to the pathologic shunts of the cavernous sinus (5–8, 10–12). Even if the IPS is partially or completely thrombosed, its recanalization and consequent transvenous occlusion of a dural CCF is often possible because of improved materials and experience in venous catheterization (7, 12, 13). If the IPS route is not possible or has failed, an anterior transvenous approach to the cavernous sinus through the SOV represents another alternative (6, 17, 20, 25, 26). Other unusual percutaneous transvenous approaches, including the superior petrosal sinus (34), the contralateral pterygoid plexus (35), and a cortical vein (36), have been described in few cases. Embolization through the surgically exposed superficial middle cerebral vein has been reported (37, 38). However, these approaches performed by using a venous puncture after a preliminary burr hole in the proximity of the dural fistula region should be considered only as last resort (14, 39).
The anterior approach to the cavernous sinus through the SOV is usually performed after surgical exposure of the SOV or of the angular vein (6, 15–25, 27). The SOV surgical route was first described by Peterson et al (40). Tress et al (15) were among the first to report this technique in one patient with a dural lesion. Later, Courtheoux et al (8, 16) and Labbé et al (41) described a case of a patient with a bilateral spontaneous dural CCF treated after exposition of both angular veins. More recently, relatively larger series of patients treated by surgical SOV approach included that presented by Monsein et al (18) and Miller et al (20) and the series of 12 patients reported by Goldberg et al (22) and Quinones et al (24). SOV exposure has been performed also in patients with posttraumatic direct CCF (42). Direct percutaneous puncture of the SOV is more rarely performed. Teng et al (17) described a new approach for transcutaneous puncture and catheterization of the SOV that was performed in four of five patients under the guidance of real-time digital subtraction angiography. Benndorf et al (26) reported a deeper percutaneous puncture of the SOV within the orbit in one patient. Direct transorbital puncture of the cavernous sinus through the superior orbital fissure has been also performed in the treatment for direct CCF (21). Komiyama et al (43) reported the case of a dural CCF for which catheterization of the SOV through the retromandibular vein, the superficial temporal vein, and then the periorbital vein after percutaneous puncture of the external jugular vein was performed.
Although the transfacial SOV approach to the cavernous sinus is mentioned by several authors, only few papers specifically dealing with this route have been published. Otherwise, the surgical direct transangular/trans-SOV procedures are sometimes reported as synonymous of a transfemoral transfacial route. Naito et al (44) reported a case of dural CCF treated after skin incision, exposure, and cannulation of the facial vein at the base of the mandible. These authors stated that incision of the mandibular region causes less psychologic pressure on the patient and that the operative scar is less conspicuous than that on the eyelid. Scott et al (45), reported five patients with dural CCF treated by direct facial vein puncture by using a dermatome below the malar eminence. This technique used a Doppler needle and a puncture kit with a dilator. Mizuno et al (46) described a case of dural CCF treated via the facial vein by an external jugular vein approach. These authors reported great difficulty in reaching the SOV from the angular vein with the microcatheter. Halbach et al (31) reported to have used a transfemoral venous access through the angular vein and the SOV to the fistula site in a dozen cases. In a recent article (11), the same authors reported a 15-year series of 133 patients with dural CCF treated by using an endovascular route. One hundred one (76%) patients underwent embolization with access to the cavernous sinus via the IPS or SOV. Also, in other reports (47, 48), these authors stated that transfemoral venous access to the cavernous sinus is usually via the IPS and, in rare instances, via the SOV in the orbit, the angular/facial vein, and the pterygoid plexus.
To the best of our knowledge, no published series detail the methods or report the results obtained by using the facial vein as a single route. We herein report our results of endovascular treatment of dural CCF via the transvenous transfemoral (in one case transjugular) approach through the facial vein.
In case of anterior fistula drainage, the visualization of the facial vein from the angular vein to the internal jugular vein can usually be appreciated and used as a road map. The facial vein joins the internal jugular vein at the level of the hyoid bone. Visualization of the facial vein during internal carotid angiography can also be observed in normal conditions if the extraocular branches of the ophthalmic artery are prominent and nasal vascularity is marked (49). For successful catheterization of the SOV via the facial vein, some anatomic variants and abnormal courses should be considered. The facial vein may terminate in the external jugular vein in 5% of people. This variation may represent a persistent communication of the primitive linguofacial vein with the secondarily developing external jugular vein (50). More uncommon variants include an abnormal coursing facial vein joining the retromandibular vein at a higher level in the parotid gland (51) and a facial vein terminating in the superficial temporal vein (52). Other authors have reported a facial vein flowing into the subclavian vein via the jugular arch without connection to the internal jugular vein (44).
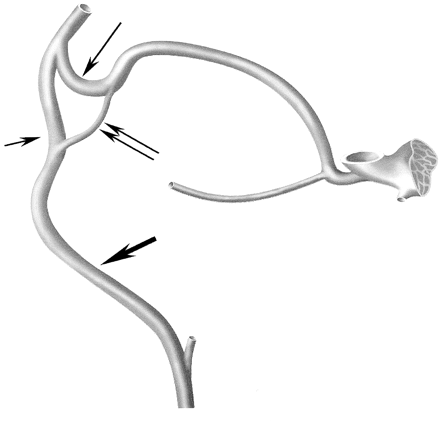
The SOV originates at the junction of its inferior and superior roots (frequently described as the inferior and superior tributaries). The inferior root, often considered as the true initial segment of the SOV, is a lateral tributary of the angular vein, whereas the superior root is a continuation of the frontal venous system (41, 53) (Figs 1 and 2). Some authors (54) consider that the initial portion of the superior root is the infratrochlear vein and that the true SOV superior root begins more deeply, at the junction of the infratrochlear vein with the supraorbital vein.
Schematic drawing of the venous system connecting the facial vein and the cavernous sinus. Facial vein (thick arrow), angular vein (short thin arrow), SOV inferior root (double arrows), SOV superior root (long thin arrow).
The orbital venous system consists of a freely anastomosing and variable network of vessels. Although this system is often reported valveless (as are the angular and facial veins), valves have been described, especially in the inferior root of the SOV (55, 56). The valves in the inferior root of the SOV have been reported to prevent flow from the facial and angular veins toward the SOV and cavernous sinus. Although the role of these valves and the flow direction are not fully understood, it is accepted that in the SOV, the venous flow is preferentially anteroposterior into the cavernous sinus.
Thanks to improved materials for microcatheters and guidewires, in all cases, we were able to catheterize the facial vein and the angular vein and to pass the tortuous roots of the SOV and their junction with the SOV. However, in two cases, it was difficult to navigate through the tortuous SOV roots, which are the most challenging segments during catheterization. Although the SOV inferior root is considered to be the origin of the SOV and it represents the more direct route, in most of our cases, we navigated through the SOV superior root. The SOV inferior root was not always visualized or it appeared a more difficult route; also, one can speculate regarding the role of a valve system. However, it must be kept in mind that in pathologic conditions, venous structures can be hardly identifiable. We experienced no difficulties during catheterization of the SOV, despite that its course is often reported tortuous, with abrupt angulations in the orbit and with a constant narrowing at the superior orbital fissure. Although catheterization of the cavernous sinus through a thrombosed SOV is possible (24, 27), in one case of a previously thrombosed SOV, we were not able to pass through.
We did not observe technical or clinical complications; however, our series of seven patients is rather small to assume general assertions. Potential complications of the transfacial approach, both hemorrhagic and thrombotic, must be considered. Some authors (31) stress the risk of damage or perforation of the SOV near the trochlea. We agree that great care must be taken during navigation through the SOV roots (usually the superior one, adjacent to the trochlea), which, in our experience, are the most difficult venous structures to pass. As reported by Uflacker et al (42), the risk of venous perforation is higher in recent CCF in which the venous wall is not yet arterialized. Among the complications of the venous approach, acute thrombosis must be kept in mind. Inagawa et al (28), in a case of direct CCF, reported an aggravation caused by acute thrombosis of the IPS during catheterization of the IPS with a 5-French catheter. Benndorf et al (26) observed partial acute thrombosis of the SOV in two patients after catheter navigation via the facial and angular veins. Catheterization of the SOV, irrespective of the approach (surgical, transfacial, frontal vein), can result in acute thrombosis. If the venous outflow is occluded without obliteration of the fistula, severe deterioration of vision and worsening of symptoms requiring emergency intervention is possible. Also, injury to the SOV may result in redirection of the venous drainage to cortical pathways, increasing the risk of neurologic complications (35). To avoid impairment or stasis of the venous outflow, we avoid distal catheterization of the facial vein with the guiding catheter, which is usually positioned in the internal jugular vein. In contrast to some authors (16, 17, 20, 24) who use material that is larger in size (4- or 5-French sheaths and/or catheters in surgical SOV approach), we prefer only small microcatheters and microwires for navigation through the SOV. In addition, all our procedures are performed under full heparinization.
The use of a transfemoral transfacial approach requires caution, and it should be reserved for selected cases. This approach is indicated in case of thrombosed IPS and when no communication exists between the compartments of the cavernous sinus involved by the CCF and the IPS itself (as observed in two of our cases). According to some authors (11, 13, 31, 57), in the treatment of dural CCF, the catheterization of the IPS represents the most direct approach and it should be attempted first. Although complications of the IPS route (including abducens nerve palsy, intracranial hemorrhage, cerebellar or extradural hematoma) have been reported in association with dural (7, 12, 28, 29) and direct (57, 58) CCF, this approach is relatively safe.
Similarly, the transfemoral route through the facial vein seems less traumatic than the SOV direct approach. It does not require cut-down or puncture of the SOV and avoids cutaneous scars, which are not always cosmetically acceptable (22, 44). Disadvantages and complications of the SOV exposure include bleeding from the SOV, difficulty in identifying the vein, injury of the supraorbital nerve and levator muscle, damage to the trochlea, infection, and granuloma (12, 22, 24). Oishi et al (12) reported bilateral forehead dysaesthesia in one patient and blepharoptosis in two of eight patients undergoing surgical exposure of the SOV. In addition, it has been reported that if the SOV is too small, tortuous, thrombosed or if it is located deep in the orbit or has anatomic variations, surgical transorbital venous access may not be possible (22). On the other hand, if a transfacial approach fails as a consequence of internal jugular vein or SOV thrombosis, spasm, and/or anatomic variations, the SOV direct approach remains a valid option. However, this later technique requires an experienced orbital surgeon who is not always available.
Conclusion
In the endovascular treatment of dural CCF, the transfemoral approach of the cavernous sinus via the facial vein provides a valuable and safe alternative, allowing effective treatment of these lesions. Based on our experience, this approach is indicated if a venous drainage toward the SOV exists, if the IPS is thrombosed, if there is no communication between the IPS and the fistula site, or if the IPS approach has failed. Catheterization of the cavernous sinus via the facial vein usually is successful. Factors that could impair the positioning of the microcatheter at the level of the CCF are specific anatomic configurations, venous thrombosis (particularly of the SOV), and trabeculae within the cavernous sinus.
Acknowledgments
We thank Dr. G. Benndorf for helpful discussion of the manuscript and for providing the schematic drawing (modified from Benndorf G, Thesis, Berlin, 2002).
References
- Received October 7, 2002.
- Accepted after revision January 6, 2003.
- Accepted after revision January 6, 2003.
- Copyright © American Society of Neuroradiology